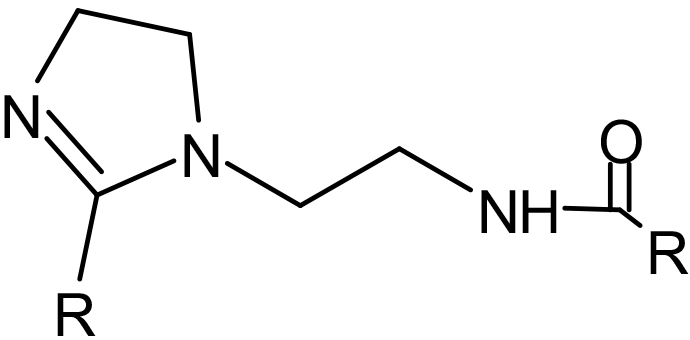
IMIDAZOLINE
Aminoethyl Imidazolines are produced by reacting DETA (Diethylene Triamine) with fatty acids (1:2) producing an amide and then cyclizing to produce a 5 member ring containing 2 nitrogen’s with an ethyl amine group attached that is further amidized. The R group attached to the ring and amide will be determined by the fatty acid used. The un-neutralized imidazoline will be lipophilic making them soluble in mineral oil or solvents and water dispersible.
Due to the presence of 2 alkyl groups on the molecule, amidoethyl imidazolines are the most oil soluble (hydrophobic) of the imidazolines. They have good film forming, with good corrosion protection and lubrication. Are acid stable.
ULTRAFAX 7220
An amidoethyl Imidazoline (1:2) of Tall oil.
Appearance: Clear amber liquid
Amine Value: 70-170 mg KOH/g
Solubility: Oil soluble and water dispersible
Min % Imidazoline: 45
Acid Value: <10 mg KOH/g
Moisture: <0.1%
APPLICATION: Used in Agriculture, lapping cpds, pigment dispersion. Acid stable, used in flocculation and softeners.
